INMETRO and ANATEL Certification: Requirements for Authorization in Brazil?
If ANVISA registration is no longer enough
As a manufacturer, there are some hurdles you have to clear before selling your medical device in Brazil. The best known is registration with ANVISA. In addition to this, you may also need INMETRO or ANATEL certification for your device.
Manufacturers should know in which cases they need to obtain INMETRO or ANATEL certification and comply with additional requirements. Otherwise, there is a risk of nasty surprises during the authorization process, for example, it may become longer, or you may incur unplanned costs.
In this article you will learn:
- Under which circumstances, which authorities other than ANVISA will inspect your devices
- How to schedule the necessary tests
Authorization in Brazil
It is a good idea to read the article on the authorization of medical devices in Brazil first. That article will also explain how to classify your medical devices in Brazil and register them with ANVISA.
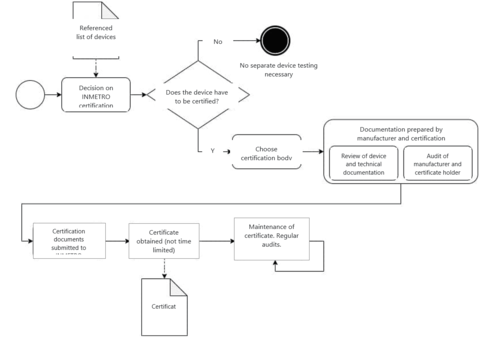
1. INMETRO and ANATEL certificates as part of the authorization documents
Some devices can only be marketed in Brazil with INMETRO or ANATEL certification. These two authorities are responsible for testing and certification.
Manufacturers may not apply for registration with ANVISA for these medical devices until they can have the INMETRO or ANATEL certificate. The certificate must be submitted to ANVISA together with the device documentation.
2. INMETRO certification
a) Who is INMETRO?
INMETRO is the National Institute of Metrology Standardization and Industrial Quality. The authority is not only responsible for medical devices, but also, more generally, for products that contribute towards the safety and – as in the case of medical devices – the health of people. So, in addition to medical devices, it is responsible for products such as domestic appliances, wheels, tires and lights.
Its testing ensures that products meet defined minimum standards. Typically, these are national versions of international standards, such as ABNT NBR IEC 60601-1:2010 + Emenda 1:2016 or ABNT NBR IEC 60601-1-2:2017.
The requirements for the INMETRO certification are defined in Resolution 384/2020. However, not every medical device is subject to testing by INMETRO.
b) Which devices are affected?
Medical devices that require an INMETRO certificate include:
- Electrical devices
- Sterile single-use syringes
- Breast implants
- Surgical and non-surgical rubber gloves
- Condoms
The complete list of products, most of which are not medical devices, can be found here.
Please note!
Whether INMETRO certification is required or not does not depend on the classification of your medical device. Certification may also be required for class I devices.
Choosing a suitable certification body
The testing and issuing of the corresponding certificate can be done by INMETRO itself or by a body recognized by INMETRO.
To apply for certification, you need to have a Brazilian company as an authorized representative. This means that if you do not have an office in Brazil, you will need a Brazilian certificate holder. This is where your authorization partner, who you planned the ANVISA registration with, comes in handy. This local partner should be able to help you select a suitable certification body.
The certification itself has two parts:
- Device verification, including a review of the technical documentation
- Audit of the manufacturer and the Brazilian INMETRO certificate holder
Certification (part 1): device verification
For the device verification, the following documents, among others, have to be submitted:
- Risk management file
- Usability engineering file
- Documentation of applicable QM and manufacturing processes
- Test reports (if available)
If your device has already been verified, it is possible to use existing test reports for the INMETRO certification. You should agree on the exact requirements with your certification body.
Test reports that meet the following conditions are often accepted:
- The report represents the current technical state of the device.
- It was written by an ILAC-certified body.
- It has been checked against the standards (and versions) recognized by INMETRO and ANVISA.
NB!
Only an ILAC-certified body can perform the device testing. Otherwise, the device testing has to be repeated.
ILAC is an international association of accreditation bodies that includes, for example, DAkkS.
You can find a list of INMETRO accredited bodies here.
Comment:
The recognized standards (versions) are listed in the normative instructions (Instrução Normativa):
If you do not yet have test reports, your device will be tested for the INMETRO certification. This can extend the certification process. And the costs of the certification will increase accordingly.
Certification (part 2): Audits
The focus of the audits is on verifying compliance with the INMETRO requirements. These include requirements for the QM system according to ISO 13485 (e.g., design and development, production) and requirements for 100% testing (e.g., leakage current or earthing according to IEC 60601-1).
The Brazilian certificate holder also has to be audited and, similar to the manufacturer audits, the focus is on auditing certain ISO 13485 criteria (including document control, non-conforming devices, corrective actions).
d) How long the INMETRO certification process lasts and what it costs
INMETRO certification can take several months. Since December 2020, the certificates issued are valid indefinitely as you long as you undergo audits to confirm that it is maintained.
Tip
Note that in order to maintain the INMETRO certificate – and, therefore, your device authorization with ANVISA – an audit by the certification body is required every 15 months. The audit is performed as part of a document review or an on-site audit.
You should budget for costs of between USD 2,500 and USD 5,000 for the maintenance of the certificate. There are also the costs of your local certificate holder on top of this.
The exact cost of the INMETRO certification depends on the corresponding certification body and the tests to be performed. Brazilian bodies often offer low-cost certification. However, they then work with auditors in the manufacturer’s home country, which means prices can vary from country to country.
3. ANATEL certification
a) Who is ANATEL?
ANATEL is the Brazilian national telecommunications authority. It demands that all telecommunications products sold in Brazil have a certificate of conformity. This obligation also applies to corresponding medical devices. As a result, an ANATEL certificate must be submitted to ANVISA for these devices.
As with the INMETRO certificate, whether an ANATEL certificate has to be submitted or not does not depend on the product class: manufacturers also require an ANATEL certificate for certain class I devices.
NB!
Note that ANATEL only recognizes Brazilian product tests. This means that you may need to have your device tested for the Brazilian market as well.
The general requirements for ANATEL certification are defined in Resolution 715/2019.
b) Which devices are affected?
All devices that have a radio module require ANATEL certification. This applies, for example, to devices with a Bluetooth or WiFi interface.
The devices affected have to undergo a conformity assessment procedure. This includes device testing in Brazil and a review of the technical documentation.
c) How to get an ANATEL certificate
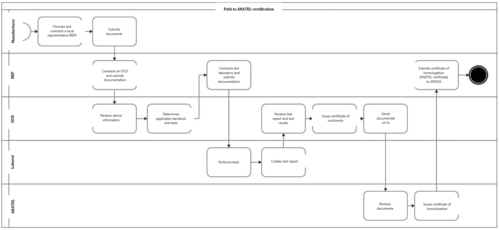
Various stakeholders are involved in the certification process:
- The manufacturer of the medical device
- A local representative (REP) in Brazil
- A test laboratory
- A certification body (OCD)
- ANATEL
In the first step, a manufacturer without an office in Brazil has to contract a local representative (REP), as only Brazilian companies can apply for ANATEL approval.
Together with the manufacturer, the local representative contracts a certification body, known as an OCD (Organismo de Certificação Designado). The OCD is responsible for identifying the standards that have to be complied with and the tests required.
To do this, it receives technical device information from the manufacturer, e.g.:
- Specifications
- Drawings
- Device labels
You can find a list of OCDs recognized by ANATEL here.
The local representative then contracts a test laboratory to carry out the necessary device testing.
Once the tests have been completed, the OCD reviews the available test reports, issues a certificate of conformity and forwards the data to ANATEL.
In the last step, ANATEL reviews the data sent and issues a certificate of homologation if the requirements have been met. This ANATEL certificate is necessary for device authorization with ANVISA.
d) How long the ANATEL process takes and what it costs
The costs and duration of the certification depends largely on the following factors:
- Availability of the test laboratories
- Length of the test
- The technology to be tested
For example, testing a device that has a Bluetooth 2.0 interface is less time-consuming and costly than testing a device with 4G technology.
Manufacturers should allow approximately six months for the entire certification process. The process includes the device verification, which can take up to three months, and the subsequent issuing of the “certificate of homologation.”
This ANATEL certificate of homologation is valid indefinitely. However, the OCD must monitor the certificates it issues. So, a review of the documentation by the OCD is required every 24 months in order to maintain the ANATEL certificate and, as a result, the ANVISA device authorization. During this review, the OCD reviews whether the specifications have remained the same or have changed. When requirements change, the OCD reviews whether the certificates it has issued are still valid. In this case, the technical documentation also has to be submitted. A new test may be required if the device has been modified.
4. Conclusion
For some devices, as the manufacturer you have to meet specific requirements and obtain an INMETRO and ANATEL certificate. Nevertheless, the hurdles to device authorization in Brazil are not insurmountable. As a foreign manufacturer you will need a local representative for the authorization. Therefore, carefully selecting this representative is probably the most important step towards successful authorization.
In addition, if you meet all the applicable requirements and know the costs involved in certifications, you will be well placed to successfully plan and achieve authorization.
If you need help obtaining authorization in Brazil, e.g., with classification, with the INMETRO and ANATEL certification or with ANVISA registration, get in touch. You can use our contact form. The Johner Institute’s experts will be happy to help. They will even do so free of charge via our micro-consulting (German) service.