Market Access of Medical Devices in Brazil
Getting medical devices registered in Brazil is not easy. This is partly due to the number of regulations and their complexity and partly due to the fact that Brazil has published most of its regulations in Portuguese only.
This article will give you an overview of how medical devices are regulated in Brazil and detail some of the similarities between the Brazilian system and the European and US systems. This will make it easier for you to understand and comply with the regulations and toavoidunnecessary rejections from the Brazilian authorities and the resultant delays in authorization.
This will enable you to get devices authorized in Brazil quicker and more easily.
1. The Brazilian legal system for medical devices
ANVISA is the authority responsible for medical devices in Brazil. Like the FDA:
- It passes laws on medical devices
- It checks medical device authorizations
- It monitors manufacturers’ QM systems to ensure they comply with the Brazilian requirements
ANVISA's requirements are have a lot of similarities with the requirements in the European Medical Device Directive (93/42/EEC) and the US Quality System Regulations (21 CFR part 820):
- Manufacturers who have a QM system that complies with 21 CFR Part 820 also largely meet the Brazilian QM system requirements.
- Just as the EU and FDA require registered local “representatives”, in Brazil a Brazilian Registration Holder is required.
- The authority divides medical devices into classes according to rules that are similar to the European rules. These classes, in turn, determine the time and effort required for the authorization. You will find out more on classification below.
In addition, Brazil has country-specific requirements, such as the requirement for an INMETRO certificate. More on that later.
2. Medical device legislation in Brazil
The table below gives you an overview of the most important Brazilian laws:
Product type | Resolution | Contents |
Medical devices | Registration, modification, extension and deregistration of medical devices | |
Medical devices | Requirements for class I and II device registration | |
In vitro diagnostic medical devices | Classification rules and requirements for registration and labeling of IVDs | |
All | Validity of registrations | |
All | Brazilian Good Manufacturing Practices | |
All | Regulates the authorization and surveillance of devices by the authority in Brazil | |
All | General law on health surveillance |
3. Classification according to RDC-185/2001
Manufacturers who want to register a medical device in Brazil first have to determine its risk class. ANVISA differentiates between classes I (low risk) to IV (high risk).
Devices are classified based on 18 rules established in Annex II of RDC-185/2001. These were largely taken from the European Medical Device Directive (MDD) and correspond to its 2001 version.
You can use the following table as a guideline for the classification of your device:
EU | Brazil |
I | I |
IIa | II |
IIb | III |
III | IV |
N.B!
Please note that for certain devices the classification cannot be transferred because the European rules have been amended. This is the case, for example, with software devices and surgically invasive devices.
With the Medical Device Regulation (MDR), Europe has introduced more alterations that mean that the classification systems are diverging. This makes it difficult to transfer the classification straight across.
4. Prerequisites for the authorization of medical devices in Brazil
Before manufacturers can submit the authorization documents to the Brazilian authorities, they must fulfill some prerequisites:
1st prerequisite: the B-GMP certificate
Manufacturers must comply with the requirements of the Brazilian Good Manufacturing Practices (B-GMP). These are largely based on the FDA's Quality System Regulations (21 CFR 820).
Manufacturers of class I and II devices must comply with these requirements, but this compliance is not verified by ANVISA. They also do not have to submit a B-GMP certificate with the authorization. In contrast, manufacturers of devices in risk classes III and IV must have a B-GMP certificate before the device can be authorized.
Tip
Please note that this certificate can only be issued after an inspection, e.g., by ANVISA. There may be long lead time before the authority conducts the audit.
ANVISA significantly simplified the procedure for obtaining the B-GMP certificate with Resolution RDC-183/2017, which was passed in October 2017:
- ANVISA recognizes MDSAP audits, provided that Brazilian requirements have been included.
- ANVISA recognizes audit reports from recognized bodies in IMDRF member states.
The submission of the above audit report can speed up the issuance of the B-GMP certificate and the authorization of your medical device in Brazil.
B-GMP certificates are valid for two years and the Brazilian Registration Holder (BRH) must submit an application for renewal at least six months before the expiry data.
The cost of a GMP certificate accounts for a large part of the authorization costs and can be up to approximately EUR 25,000.
2nd prerequisite: the Brazilian Registration Holder (BRH)
Another prerequisite for authorization in Brazil is a regional representative on the ground, the Brazilian Registration Holder (BRH). The BRH must be officially named and is legally responsible for the authorized device in Brazil.
This task can be performed by a company office in Brazil or a distributor. Independent companies can also be BRHs.
N.B!
Choose your BRH carefully as they will hold the registration! This means you are dependent on them.
The tasks of the Brazilian Registration Holder include:
- Communicating with ANVISA
- Applying for the B-GMP certificate
- Submitting authorization documents
- Post-market surveillance (at least, it is responsible for it)
- Vigilance: it reports recalls and incidents to ANVISA
3rd prerequisite: the INMETRO certificate
INMETRO is the National Institute of Metrology Standardization and Industrial Quality. Some medical devices require a certificate confirming that the device complies with applicable Brazilian standards and requirements before they can be authorized by ANVISA.
The testing can be performed (and the corresponding certificate issued) by INMETRO itself or by a body recognized by INMETRO. The final certificate does not expire, but requires regular audits in order to maintain its validity.
Medical devices that require an INMETRO certificate include:
- Electrical devices
- Sterile single-use syringes
- Breast implants
- Surgical and non-surgical rubber gloves
- Condoms
The complete list of products, most of which are not medical devices, can be accessed here.
5. Registration process
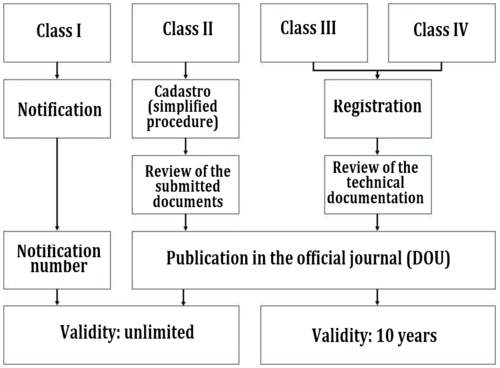
ANVISA has two different registration pathways. They differ according to the classification:
- Class I and II devices are notified to ANVISA and then given a notification number.
- Class III and IV devices go through a complete authorization and testing procedure.
6. Documentation requirements
The class and, therefore, the registration procedure also determine the documentation requirements. The higher the class of your device, the more extensive the documentation requirements are.
a) Requirements that apply to all classes
Irrespective of the class of the device, manufacturers must meet the (documentation) requirements set out in Annexes III A, III B and III C of RDC-185/2001:
Annex | Content |
III A | Form with general information on the device, the manufacturer and the type of authorization (this will be submitted to ANVISA by your BRH). |
III B | Requirements for labeling and instructions for use:
|
III C | Requirements for the technical report |
In addition, manufacturers must have proof of:
- Compliance with technical requirements (where applicable, in the form of an INMETRO certificate)
- Confirmation of payment of the authorization fees
b) Requirements for the registration of class I and II medical devices
Class I and II products are registered through a simplified notification process. In addition to the above mentioned requirements, manufacturers submit the authorization of the BRH and a confirmation of compliance with B-GMP requirements.
ANIVSA will issue a notification number without reviewing the technical documentation.
However, ANVISA reserves the right to request further documentation.
The registration of class I and II products does not expire.
N.B!
Please note that if ANVISA does carry out a review, you will have to make your technical documentation available to the BRH.
c) Requirements for the registration of class III and IV medical devices
For class III and IV medical devices, ANVISA carries out a detailed review of the documentation. The authority requires manufacturers to submit a large parts of their technical documentation in addition to the aforementioned documents. The documentation requested includes the:
- Device description
- Product requirements
- Information on risk management
- Software / firmware description
- Clinical evaluation
- Biocompatibility, electrical safety and electromagnetic compatibility testing (as applicable)
- Proof of usability
ANVISA will review the submitted documentation and publish it in the DOU (Diário Oficial da União) upon approval. You will not receive an additional certificate.
Authorizations issued for class III and IV devices are valid for 10 years.
d) Requirements for the registration of special medical devices in Brazil
For some devices, financial information also has to be submitted. The devices affected by this requirement include orthopedic and cardiovascular devices.
The full list can be found in Decision RE no. 3385/2006 and on ANVISA's website. You can also find a helpful template for your business report there.
The financial information required includes basic information about the device, as well as financially relevant information, such as pricing, sales figures or the estimated number of patients to be treated.
7. Conclusion and summary
The requirements for authorization in Brazil are just as extensive as those in Europe and the USA. Most of the documents can be reused by companies, but they have to be translated into Portuguese. In addition, there are some requirements that are specific to Brazil that have to be met.
As in Europe and the USA, the class of the medical device determines the duration and cost of the authorization.
| Class I | Class II | Class III | Class IV |
Complexity | Low | Low | High | High |
Duration | 1 month | 1 month | approx. 12 months | approx. 12 months |
Validity of the authorization | Unlimited | Unlimited | 10 years | 10 years |
B-GMP certificate | Not required | Not required | Yes - valid for 2 years | Yes - valid for 2 years |
The Johner Institute’s team will be happy to help you with the authorization of your medical devices in Brazil.