Vital physiological processes - decisive for classification
Both the Medical Device Regulation (MDR) and the Medical Device Directive (MDD) classify medical devices according to whether they diagnose or monitor vital physiological processes. Unfortunately, both regulations fail to define the term “vital physiological processes”. As a result, we get:
- Disputes between different manufacturers and with notified bodies
- Resulting lawsuits
This article provides the missing definition.
1. Vital physiological processes and classification rules
a) MDR Rule 10
Rule 10 of the Medical Device Regulation (MDR) states:
“Active devices intended for diagnosis and monitoring are classified as class IIa, [...] if they are intended to allow direct diagnosis or monitoring of vital physiological processes, unless they are specifically intended for monitoring of vital physiological parameters and the nature of variations of those parameters is such that it could result in immediate danger to the patient, for instance variations in cardiac performance, respiration, activity of the central nervous system, or they are intended for diagnosis in clinical situations where the patient is in immediate danger, in which cases they are classified as class IIb.”
Medical Device Regulation (MDR), Rule 10
This classification rule gives some examples of vital physiological processes.
b) MDR Rule 11
Rule 11 also refers to vital physiological parameters:
“Software intended to monitor physiological processes is classified as class IIa, except if it is intended for monitoring of vital physiological parameters, where the nature of variations of those parameters is such that it could result in immediate danger to the patient, in which case it is classified as class IIb.”
Medical Device Regulation (MDR), Rule 11
c) MDD Rule 10
Rule 10 of the Medical Device Directive (MDD) is almost exactly the same as Rule 10 of the MDR:
“Active devices intended for diagnosis are in Class IIa, [...] if they are intended to allow direct diagnosis or monitoring of vital physiological processes, unless they are specifically intended for monitoring of vital physiological parameters, where the nature of variations is such that it could result in immediate danger to the patient, for instance variations in cardiac performance, respiration, activity of CNS in which case they are in Class IIb.”
Medical Device Directive (MDD), Rule 10
2. The issue
People are increasingly arguing in court about what makes a bodily function - a physiological process - ‘vital’ and what is meant by a physiological process.
Medical devices have to be classified in different ways based on these decisions.
Further information
Read more on the classification of medical devices here.
3. Working out what vital physiological processes are
As the authors of the MDR and MDD have failed to define the term “vital physiological processes”, we have to search for other sources.
a) Global Harmonization Task Force (GHTF)
The GHTF has published a document with the title “Principles of Medical Devices Classification” and number “GHTF/SG1/N77:2012”.
In it, it briefly and precisely defines the term “vital physiological process” used by the MDR:
Definition: Vital physiological process
“Vital physiological process means a process that is necessary to sustain life, the indicators of which may include any one or more of the following:
- respiration;
- heart rate;
- cerebral function;
- blood gases;
- blood pressure;
- body temperature.”
This should be an adequate answer to the question. In one court case the judge followed a similar argument.
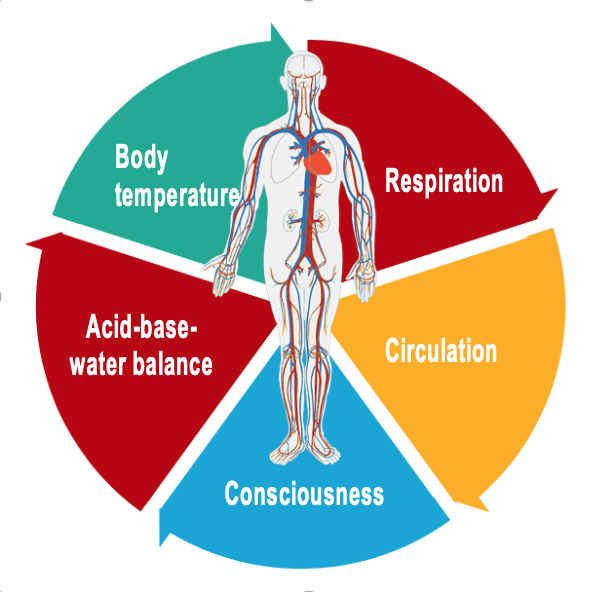
Fig. 1: Overview of vital physiological processes
b) Wikipedia
Wikipedia refers to an article on “vital signs”. There it says:
“Vital signs (often shortened to just vitals) are a group of the 4 to 6 most important signs that indicate the status of the body’s vital (life-sustaining) functions. These measurements are taken to help assess the general physical health of a person, give clues to possible diseases, and show progress toward recovery. The normal ranges for a person’s vital signs vary with age, weight, gender, and overall health.
There are four primary vital signs: body temperature, blood pressure, pulse (heart rate), and breathing rate (respiratory rate), often notated as BT, BP, HR, and RR.”
c) Pschyrembel
The Pschyrembel dictionary defines vital processes as follows:
Definition: Vital processes
“Body functions that ensure the organism's life processes. In a strict sense, these are respiration and cardiovascular function, in the broader sense, they include brain function (consciousness) and so-called secondary vital processes, which include the water-electrolyte and acid-base balance, kidney function and heat balance.”
Source: Pschyrembel
This definition obviously distinguishes between first order and second order vital processes. As the water-electrolyte and base balance are closely related to blood gases, we can see that this broadly agrees with the physiological processes specified by the GHTF.
d) Reference text books
The “door-stop” Innere Medizin by Classen et al. writes:
“Life-threatening vital dysfunctions are failures of respiration, cardiovascular function, brain function, regulation of the water-electrolyte-acid-base balance, including kidney function (homeostasis), blood coagulation and fibrinolytic system (hemostasis), temperature regulation and metabolic regulation.”
Innere Medizin by Classen/Diehl/Kochsieck
Blood coagulation and metabolic regulation are not included in the GHTF's list.
e) Summary
Physiological process | GHTF | Wikipedia | Pschyrembel | Classen |
Respiration | X | X | X | X |
Cardiovascular system: pulse, blood pressure | X | X | X | X |
Brain, consciousness | X | X | X | |
Blood gases and electrolyte balance | X | X | X | |
Body temperature | X | X | X | |
Blood coagulation | X | |||
Metabolic regulation | X |
Therefore, the following physiological processes are not vital physiological processes:
- Senses: Sight, hearing, smell, touch, taste
- Reflexes e.g. patellar tendon reflex, corneal reflex
- Spatial orientation
4. Conclusion
There is no conclusive and unambiguous definition of what is meant by a vital physiological process. But for the question of what makes a physiological process vital one in order to classify a medical device accordingly, the GHTF has given a clear answer.
It is not very productive to regularly try to argue in court that other physiological functions should also be counted as vital physiological functions. This leads to an unnecessarily high classification and an additional burden for manufacturers, while not providing any additional safety for patients.
If a case does come to court, please contact us or one of our specialized lawyers. We recommend the following:
- Dr. Lücker http://www.mp-recht.de/
- Prof. Dr. med. Dr. iur. Christian Dierks from Dierks + Company


