Intended Use and the Usage According to Regulations
Intended Use (check out also GHTF/SG1/N70:2011) is an objective aim of medical products manufacturers related to the purpose of the products, processes or service. It is about how the specification, instructions and informations of products, processes or service, ensure the purpose of it.
Definition of the term intended use (of medical devices)
The term intended use includes medical devices, strictly speaking in two aspects:
- The actual medical purpose, that is, which disease or injury you can diagnose, treat or monitor
- The authorised use, including the intended use, but also other planned actions of the user with this medical device such as storage, transport, update or cleaning.
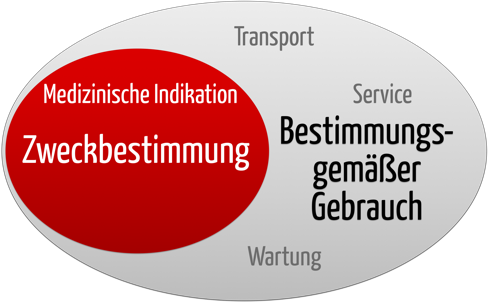
Die IEC 60601-1:2013 defines the "usage in accordance to regulations" as operation, including routine inspection and adjustments through user, and standby correspondent to instruction manual.
Elements of a intended use
Address the following aspects in your intended use:
- Medical indication (for example, illness)
- Intended patient group
- Probable body part
- Probable User Profile
- Intended use environment / usage environment
- Functioning, physical principle
- Other Intended use
The training videos in auditgarant will show you how you can document a intended use and comply with the law and still be able to condense documents. As a premium member of the auditgarant even a template with sample content is available, you only need to adjust it to your medical device.
